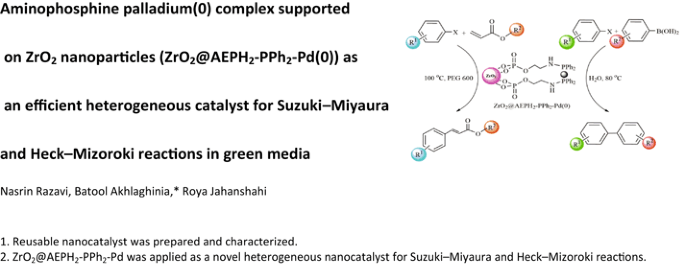
Aminophosphine Palladium(0) Complex Supported on ZrO2 Nanoparticles (ZrO2@AEPH2-PPh2-Pd(0)) as an Efficient Heterogeneous Catalyst for Suzuki–Miyaura and Heck–Mizoroki Reactions in Green Media | SpringerLink

Proposed palladium(II)/palladium(IV) catalytic pathway for the C(sp 3... | Download Scientific Diagram

Synthesis and catalytic application of Pd complex catalysts: Atom‐efficient cross‐coupling of triarylbismuthines with haloarenes and acid chlorides under mild conditions - Jadhav - 2017 - Applied Organometallic Chemistry - Wiley Online Library
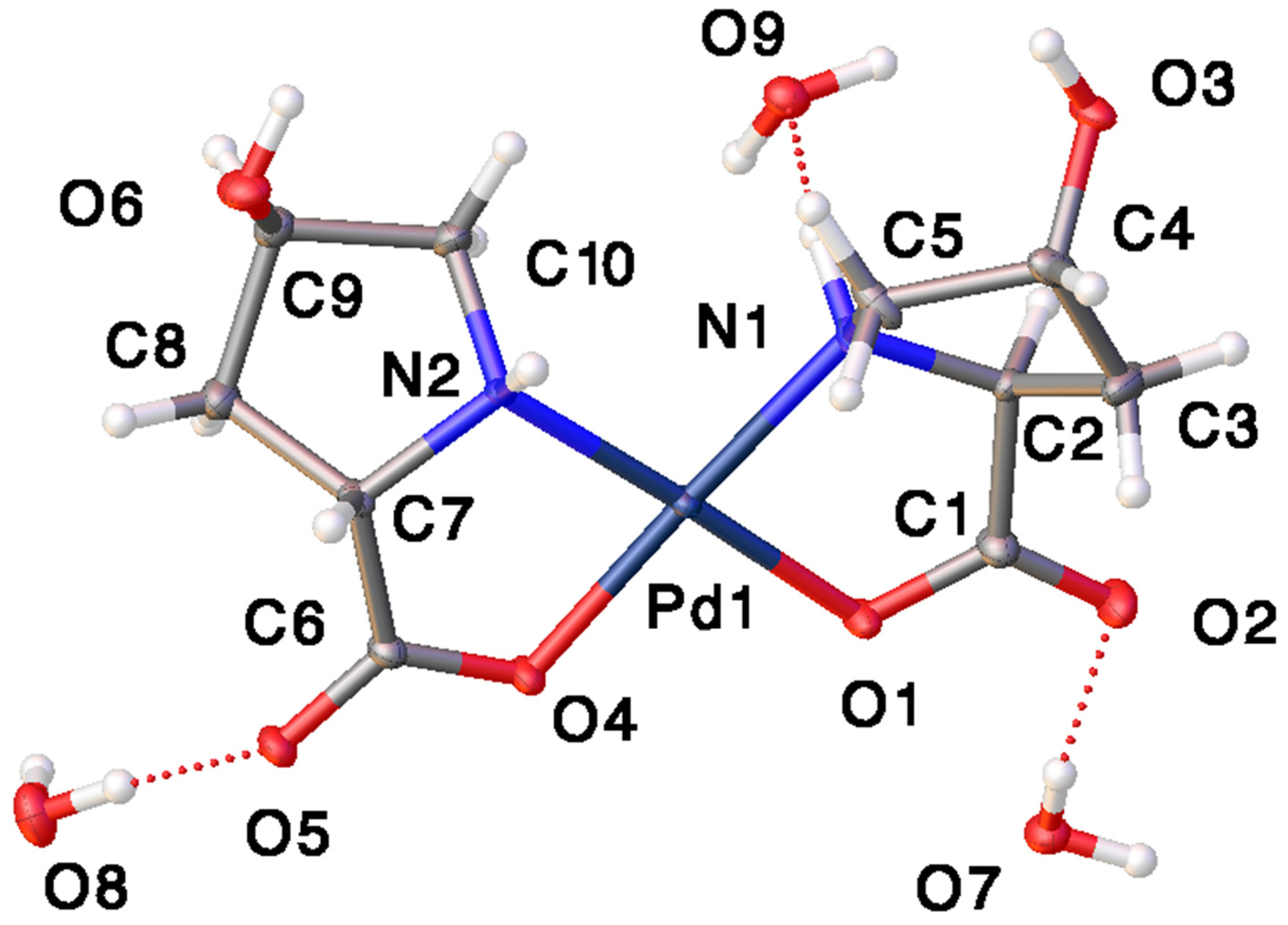
Catalysts | Free Full-Text | Synthesis, Structure, and Catalytic Reactivity of Pd(II) Complexes of Proline and Proline Homologs | HTML
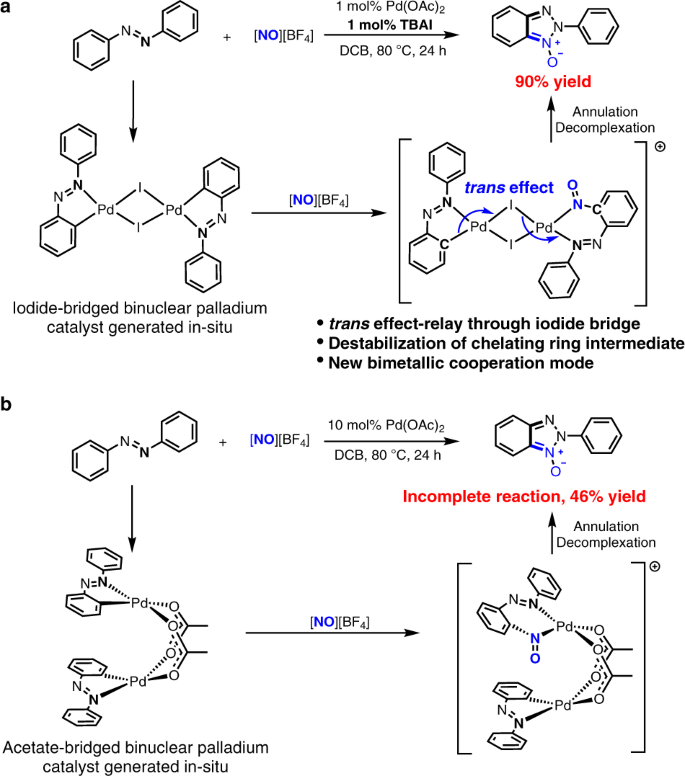
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
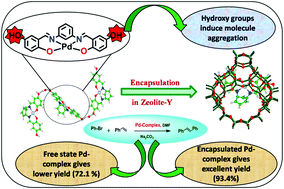
An insight into the catalytic activity of palladium Schiff-base complexes towards the Heck coupling reaction: routes via encapsulation in zeolite-Y - Dalton Transactions (RSC Publishing)

A Robust Palladium(II)–Porphyrin Complex as Catalyst for Visible Light Induced Oxidative CH Functionalization - To - 2013 - Chemistry – A European Journal - Wiley Online Library

Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML
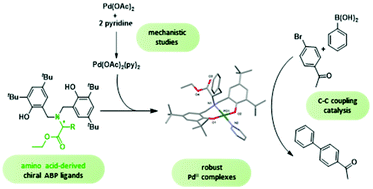
Amino acid-derived bisphenolate palladium complexes as C–C coupling catalysts - Dalton Transactions (RSC Publishing)
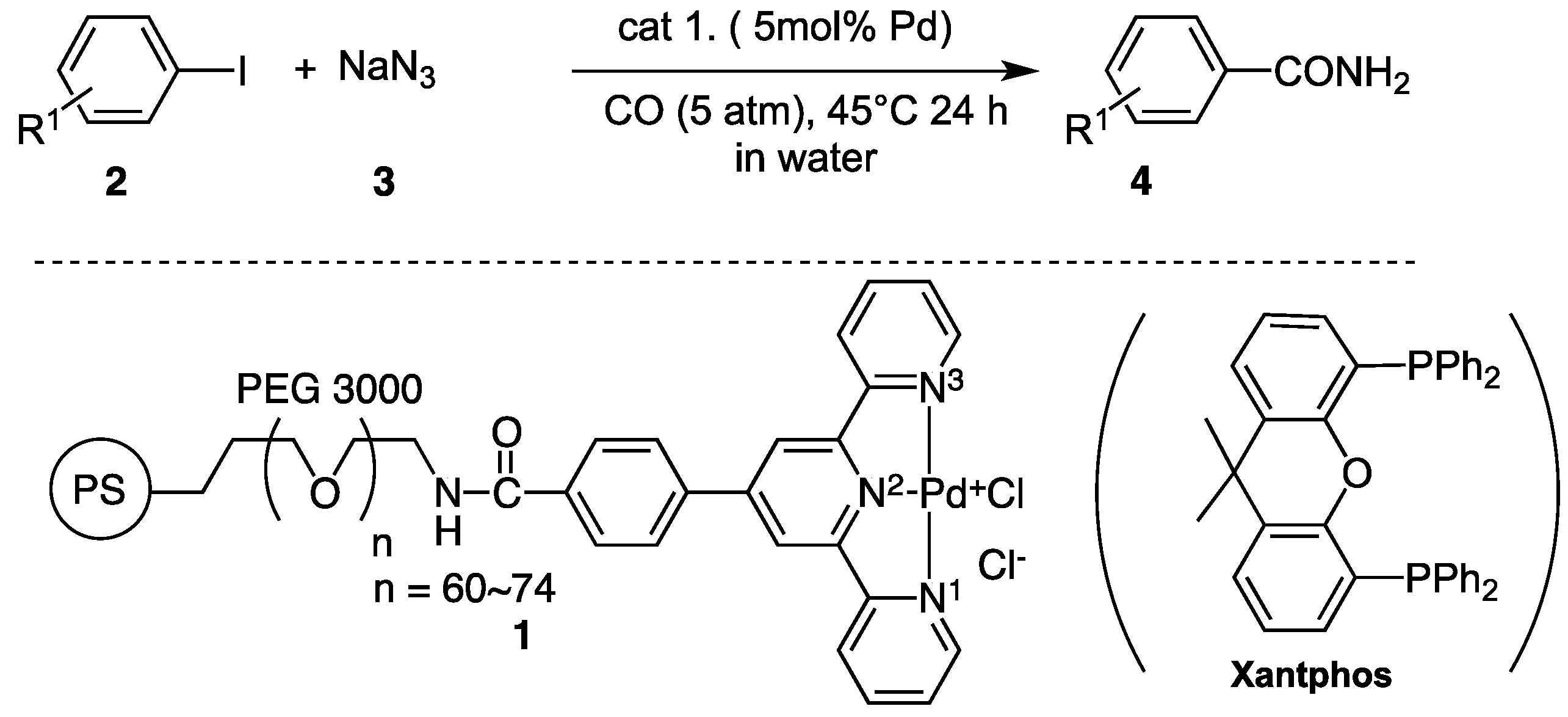
Catalysts | Free Full-Text | Recyclable Polymer-Supported Terpyridine–Palladium Complex for the Tandem Aminocarbonylation of Aryl Iodides to Primary Amides in Water Using NaN3 as Ammonia Equivalent | HTML

Palladium complexes bearing pyridylthioether ligands. Synthesis and application as efficient phosphine-free catalysts in Suzuki-Miyaura couplings - ScienceDirect

Palladium(II)-pivaloyl thiourea complexes: Synthesis, characterisation and their catalytic activity in mild Sonogashira cross-coupling reaction - ScienceDirect

Di-Palladium Complexes are Active Catalysts for Mono-N-Protected Amino Acid Accelerated Enantioselective C-H Functionalization | Catalysis | ChemRxiv | Cambridge Open Engage

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | SpringerLink

Asymmetric allylic substitution by chiral palladium catalysts: Which is more reactive, major π-allyl Pd(II) species or minor π-allyl species? - ScienceDirect
![Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | Tokyo Chemical Industry UK Ltd. Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | Tokyo Chemical Industry UK Ltd.](https://www.tcichemicals.com/assets/cms-images/03.gif)
Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | Tokyo Chemical Industry UK Ltd.
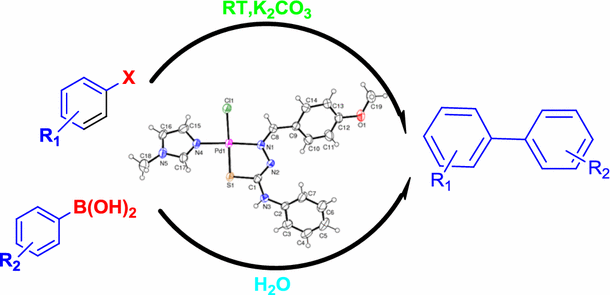
A thiosemicarbazone–palladium(II)–imidazole complex as an efficient pre- catalyst for Suzuki–Miyaura cross-coupling reactions at room temperature in aqueous media | SpringerLink
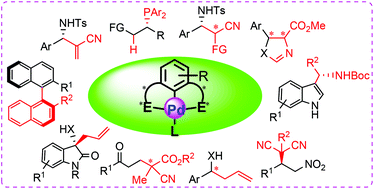
Chiral palladium pincer complexes for asymmetric catalytic reactions - Organic & Biomolecular Chemistry (RSC Publishing)

Proposed catalytic cycle for the Pd/L2-catalyzed methoxycarbonylation... | Download Scientific Diagram

Catalytic cycle of the Pd-promoted carbon-carbon (C-C) bondformation... | Download Scientific Diagram





