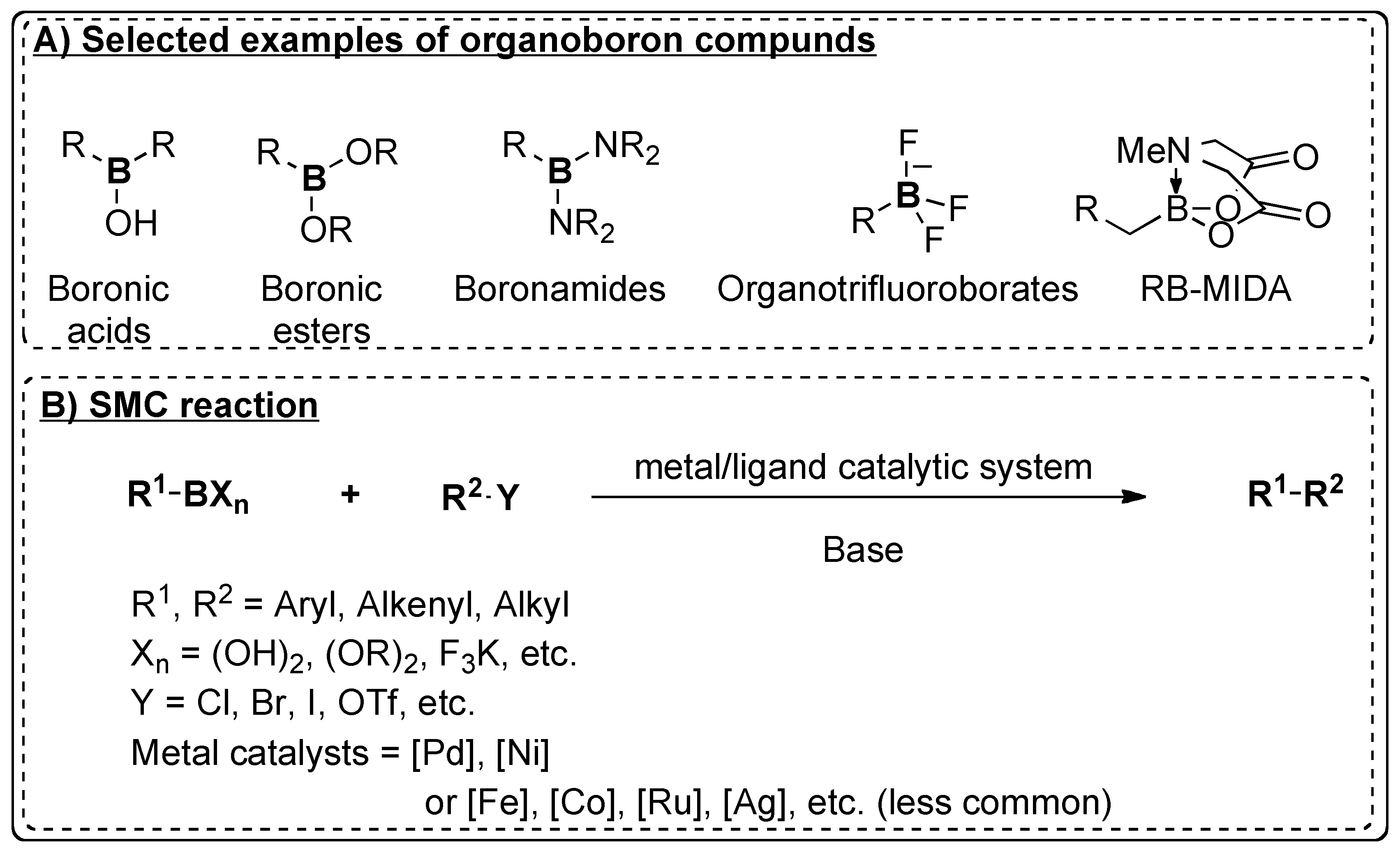
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML

Ligand-Free Suzuki–Miyaura Coupling Reactions Using an Inexpensive Aqueous Palladium Source: A Synthetic and Computational Exercise for the Undergraduate Organic Chemistry Laboratory – topic of research paper in Chemical sciences. Download scholarly ...
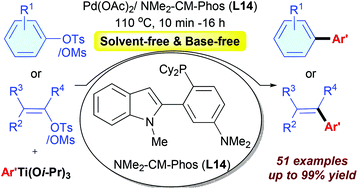
Palladium-catalyzed cross-coupling of (hetero)aryl or alkenyl sulfonates with aryl titanium as the multi-functional reagent - Organic Chemistry Frontiers (RSC Publishing)

SciELO - Brasil - Pd-catalyzed Suzuki cross-Coupling reaction of bromostilbene: insights on the nature of the boron Species Pd-catalyzed Suzuki cross-Coupling reaction of bromostilbene: insights on the nature of the boron Species

A New Ketone Synthesis by Palladium-Catalyzed Cross-Coupling Reactions of Esters with Organoboron Compounds

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect

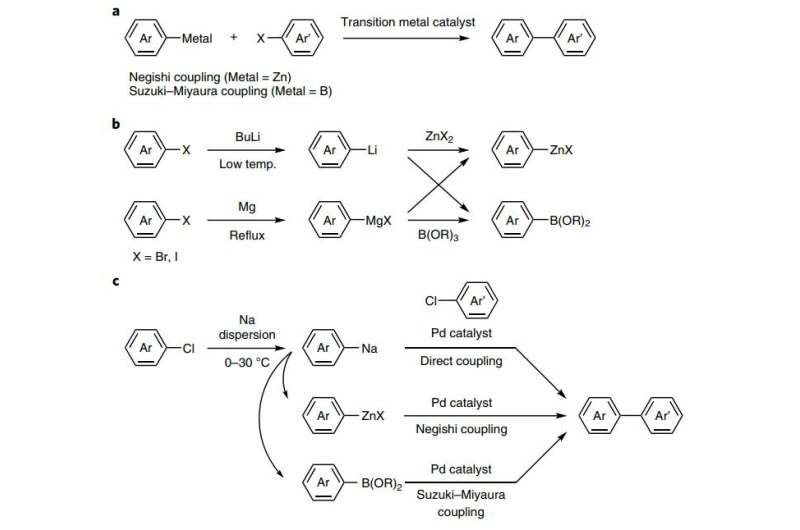
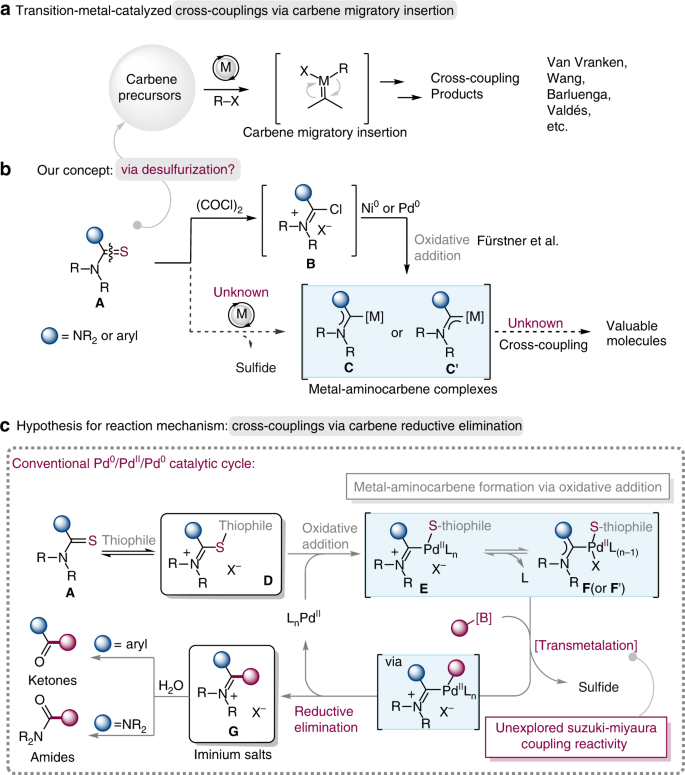
Stereoselectivity in Pd-catalysed cross-coupling reactions of enantioenriched nucleophiles | Nature Reviews Chemistry


![PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8451ced61b613a32318b99751aaa20fcc859f274/7-Figure2-1.png)



![PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8451ced61b613a32318b99751aaa20fcc859f274/7-Table2-1.png)

![PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8451ced61b613a32318b99751aaa20fcc859f274/6-Table1-1.png)




![PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar PDF] Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8451ced61b613a32318b99751aaa20fcc859f274/11-Table3-1.png)
