Diastereoconvergent Negishi cross-coupling using functionalized cyclohexylzinc reagents. | Semantic Scholar

Electroreductive Nickel‐Catalyzed Thiolation: Efficient Cross‐Electrophile Coupling for C−S Formation - Ang - 2021 - Chemistry – A European Journal - Wiley Online Library
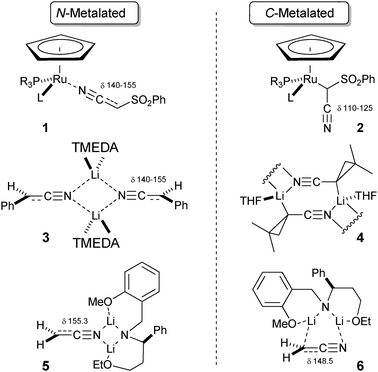
Metalated nitriles : N- and C-coordination preferences of Li, Mg, and Cu cations - Chemical Communications (RSC Publishing) DOI:10.1039/C3CC41703D

Nickel-catalyzed reductive thiolation and selenylation of unactivated alkyl bromides | Nature Communications

Electroreductive Nickel‐Catalyzed Thiolation: Efficient Cross‐Electrophile Coupling for C−S Formation - Ang - 2021 - Chemistry – A European Journal - Wiley Online Library