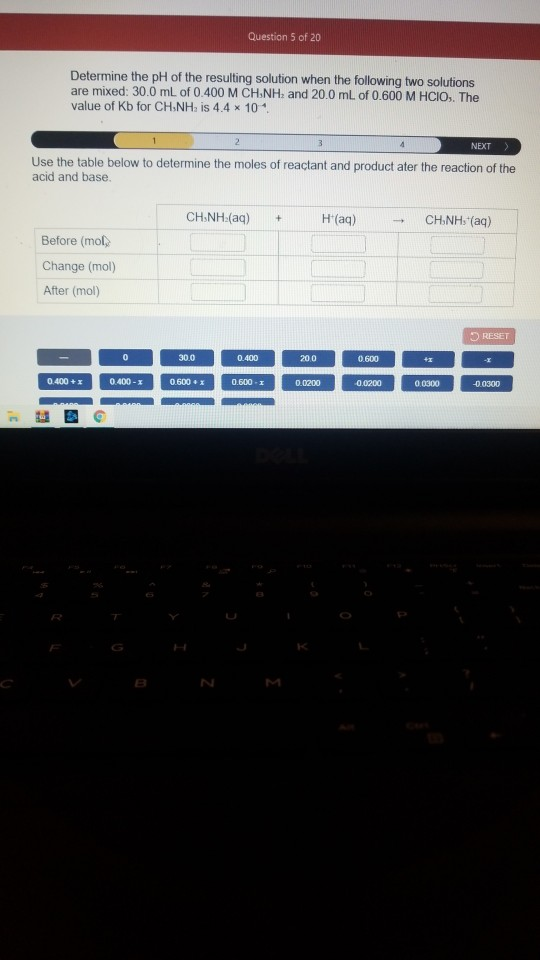
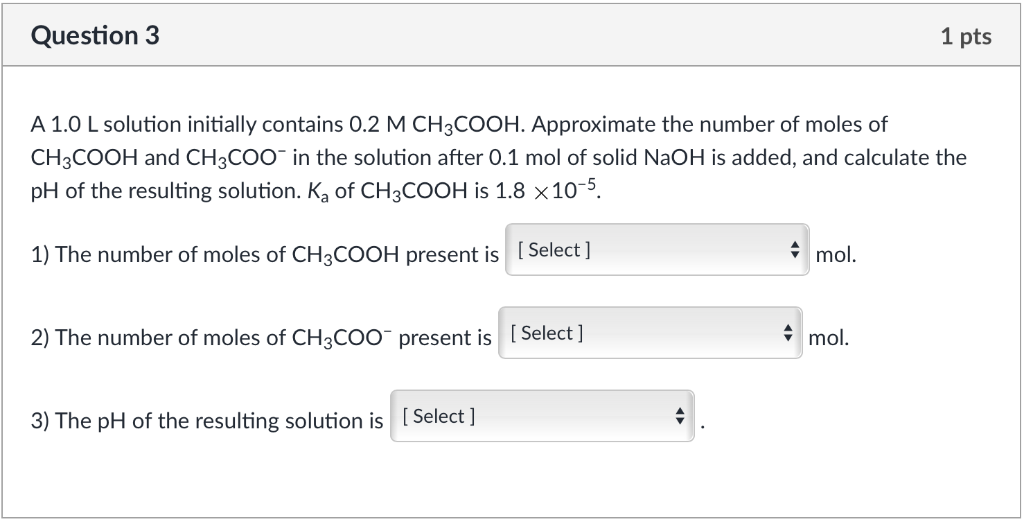
SOLVED:A buffer solution is prepared as described in a) below: A solution of a strong base is then added to this buffer solution as described in part b)_ Note: the Ka of

SOLVED:Solution 5: Mix equal volumes (10-mL each) of solution 3 (KOH) u1d solution (HCI}: Take this solution YQuT Instnctor t0 measure Ile pH Write the balanced chetuical reaction that occurs wlieu tlese

Calculate the ph of a solution formed by mixing equal volumes of two solutions A and B of a strong acids having ph=6" and "ph=4 respectively.

Equal volumes of two HCl solutions of pH = 3 and pH = 5 were mixed. What is the pH of the resulting solution?
Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively. - Sarthaks

Equal volume of two HCL solution of Ph = 3 and Ph=5 where mixed. What is the Ph of the resulting - Brainly.in

The pH of the solution obtained by mixing equal volumes of solution of pH = 5 and pH = 3 of the same electrolyte is:

Q. 16. Calculate the pH of a solution formed by mixing equal volume of two solutions A and 8 of strong acids having pH-6 and pH-4 respectively.

SOLVED:buffer solution is prepared as described in a) below: A solution of a strong base is then added to this buffer solution as described in part b): Note: the Ka of benzoic

54) The pH solution of obtained by mixing equal volumes of two solution of pH = 3 and pH = - Chemistry - Equilibrium - 10942093 | Meritnation.com
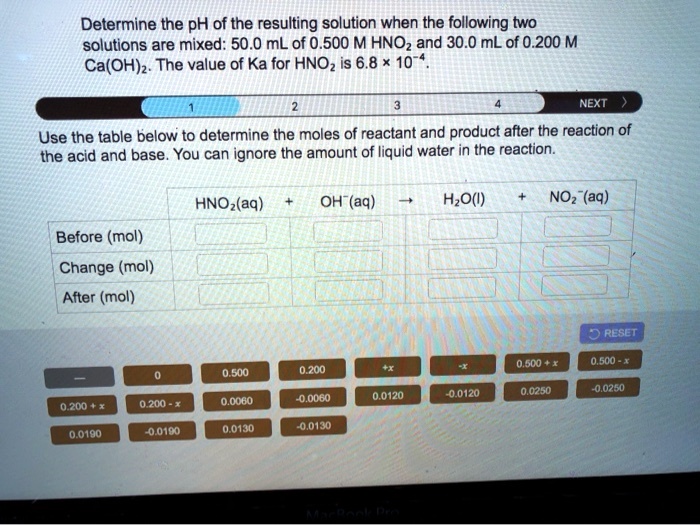
SOLVED:Determine the pH of the resulting solution when the following two solutions are mixed: 50.0 mL of 0.500 M HNOz and 30.0 mL of 0.200 M Ca(OH)z: The value of Ka for

1 Acid-base Theory 1.Arrhenius (1880s) : - Applied to aqueous solutions only Acids : Substances that produce hydrogen ions, H + (aq) when dissolved in. - ppt download